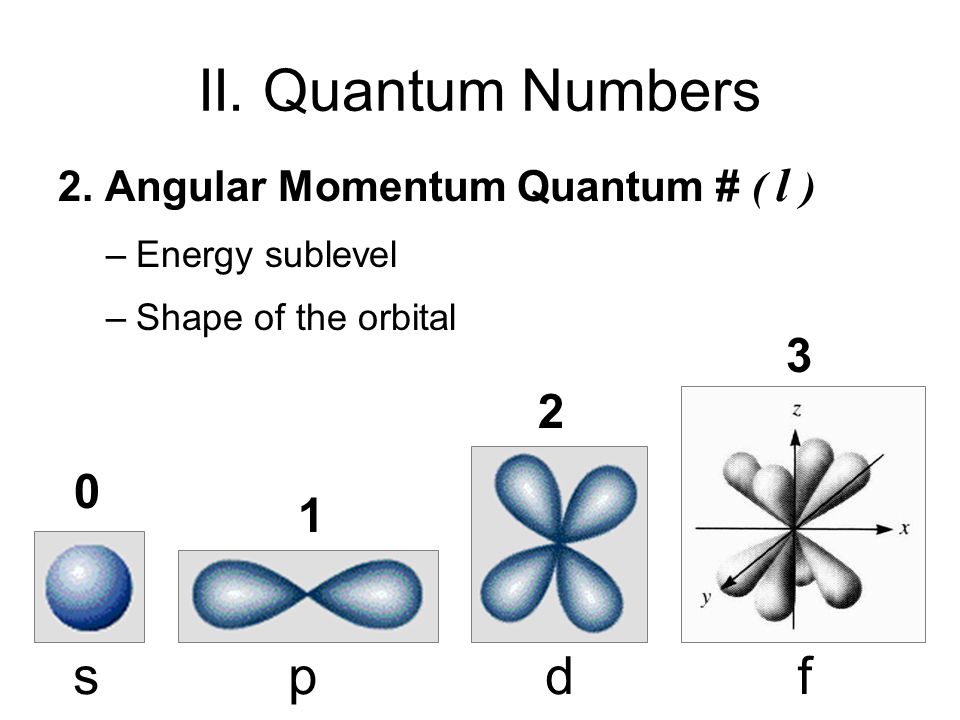
What Are The 4 Types Of Quantum Numbers . in atoms, there are a total of four quantum numbers: the four quantum numbers. the quantum numbers are parameters that describe the distribution of electrons in the atom, and therefore its fundamental. there are four quantum numbers that we are going to discuss: The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. the main quantum numbers assigned to electrons are the principal quantum number (n), angular momentum. there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. This is the same as the energy level of the.
from socratic.org
This is the same as the energy level of the. The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. in atoms, there are a total of four quantum numbers: the quantum numbers are parameters that describe the distribution of electrons in the atom, and therefore its fundamental. there are four quantum numbers that we are going to discuss: the four quantum numbers. the main quantum numbers assigned to electrons are the principal quantum number (n), angular momentum. there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers.
What are the four quantum numbers in chemistry? Socratic
What Are The 4 Types Of Quantum Numbers the quantum numbers are parameters that describe the distribution of electrons in the atom, and therefore its fundamental. there are four quantum numbers that we are going to discuss: The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. the four quantum numbers. there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. This is the same as the energy level of the. in atoms, there are a total of four quantum numbers: the main quantum numbers assigned to electrons are the principal quantum number (n), angular momentum. the quantum numbers are parameters that describe the distribution of electrons in the atom, and therefore its fundamental.
From byjus.com
Quantum Numbers Types of Quantum Numbers Chemistry Byju's What Are The 4 Types Of Quantum Numbers there are four quantum numbers that we are going to discuss: The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. the main quantum numbers assigned to electrons are the principal quantum number (n), angular momentum. This is the same. What Are The 4 Types Of Quantum Numbers.
From socratic.org
What are the four quantum numbers? + Example What Are The 4 Types Of Quantum Numbers the four quantum numbers. This is the same as the energy level of the. the main quantum numbers assigned to electrons are the principal quantum number (n), angular momentum. there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. in atoms, there are a total of four quantum numbers: there are four. What Are The 4 Types Of Quantum Numbers.
From www.w3schools.blog
Quantum Numbers W3schools What Are The 4 Types Of Quantum Numbers The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. in atoms, there are a total of four quantum numbers: the four quantum numbers. This is the same as the energy level of the. the main quantum numbers assigned to electrons are the principal quantum number (n), angular momentum. there are four. What Are The 4 Types Of Quantum Numbers.
From en.ppt-online.org
Atomic structure and properties. (Chapter 3) online presentation What Are The 4 Types Of Quantum Numbers the quantum numbers are parameters that describe the distribution of electrons in the atom, and therefore its fundamental. there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. there are four quantum numbers that we are going to discuss: The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. This. What Are The 4 Types Of Quantum Numbers.
From general.chemistrysteps.com
Quantum Numbers Chemistry Steps What Are The 4 Types Of Quantum Numbers the four quantum numbers. the main quantum numbers assigned to electrons are the principal quantum number (n), angular momentum. in atoms, there are a total of four quantum numbers: there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. there are four quantum numbers that we are going to discuss: the. What Are The 4 Types Of Quantum Numbers.
From www.pinterest.com
Quantum Numbers Explained! Quantum, Explained, Numbers What Are The 4 Types Of Quantum Numbers The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. This is the same as the energy level of the. there are four quantum numbers that we are going to discuss: there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. the main quantum numbers assigned to electrons are the. What Are The 4 Types Of Quantum Numbers.
From www.slideserve.com
PPT Quantum Numbers and Electronic Configuration PowerPoint What Are The 4 Types Of Quantum Numbers the quantum numbers are parameters that describe the distribution of electrons in the atom, and therefore its fundamental. there are four quantum numbers that we are going to discuss: The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. the four quantum numbers. there are four quantum numbers, namely, principal, azimuthal, magnetic. What Are The 4 Types Of Quantum Numbers.
From www.slideserve.com
PPT Quantum Numbers PowerPoint Presentation ID2135857 What Are The 4 Types Of Quantum Numbers in atoms, there are a total of four quantum numbers: This is the same as the energy level of the. The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. the main quantum numbers assigned to electrons are the principal quantum number (n), angular momentum. there are four quantum numbers, namely, principal, azimuthal,. What Are The 4 Types Of Quantum Numbers.
From byjus.com
Quantum Numbers (Principal, Azimuthal, and Spin) Definition What Are The 4 Types Of Quantum Numbers in atoms, there are a total of four quantum numbers: there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. This is the same as the energy level of the. the main quantum numbers assigned to electrons are the principal. What Are The 4 Types Of Quantum Numbers.
From learningschooltinkovair3.z4.web.core.windows.net
Quiz For Quantum Numbers What Are The 4 Types Of Quantum Numbers the quantum numbers are parameters that describe the distribution of electrons in the atom, and therefore its fundamental. The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. This is the same as the energy level of the. there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. in atoms,. What Are The 4 Types Of Quantum Numbers.
From nuclearchem101.weebly.com
QuatumNumbers Nuclear Chemistry What Are The 4 Types Of Quantum Numbers the quantum numbers are parameters that describe the distribution of electrons in the atom, and therefore its fundamental. the main quantum numbers assigned to electrons are the principal quantum number (n), angular momentum. the four quantum numbers. there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. there are four quantum numbers. What Are The 4 Types Of Quantum Numbers.
From www.slideserve.com
PPT Atomic Structure PowerPoint Presentation, free download ID5937240 What Are The 4 Types Of Quantum Numbers This is the same as the energy level of the. the four quantum numbers. in atoms, there are a total of four quantum numbers: there are four quantum numbers that we are going to discuss: there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. The principal quantum number (n), the orbital angular. What Are The 4 Types Of Quantum Numbers.
From www.slideserve.com
PPT Quantum Numbers PowerPoint Presentation, free download ID6766216 What Are The 4 Types Of Quantum Numbers This is the same as the energy level of the. the quantum numbers are parameters that describe the distribution of electrons in the atom, and therefore its fundamental. in atoms, there are a total of four quantum numbers: the main quantum numbers assigned to electrons are the principal quantum number (n), angular momentum. the four quantum. What Are The 4 Types Of Quantum Numbers.
From study.com
Quantum Numbers on the Periodic Table Definition & Overview Lesson What Are The 4 Types Of Quantum Numbers there are four quantum numbers that we are going to discuss: the four quantum numbers. The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. the main quantum numbers assigned to electrons are the principal quantum number (n), angular momentum. the quantum numbers are parameters that describe the distribution of electrons in. What Are The 4 Types Of Quantum Numbers.
From www.learninsta.com
Quantum Numbers What Are The 4 Types Of Quantum Numbers the main quantum numbers assigned to electrons are the principal quantum number (n), angular momentum. the quantum numbers are parameters that describe the distribution of electrons in the atom, and therefore its fundamental. there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. the four quantum numbers. there are four quantum numbers. What Are The 4 Types Of Quantum Numbers.
From www.slideserve.com
PPT Quantum Theory PowerPoint Presentation, free download ID1185278 What Are The 4 Types Of Quantum Numbers the quantum numbers are parameters that describe the distribution of electrons in the atom, and therefore its fundamental. the four quantum numbers. The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. there are four quantum numbers that we are going to discuss: there are four quantum numbers, namely, principal, azimuthal, magnetic. What Are The 4 Types Of Quantum Numbers.
From www.slideserve.com
PPT Quantum Numbers PowerPoint Presentation, free download ID3181772 What Are The 4 Types Of Quantum Numbers in atoms, there are a total of four quantum numbers: there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. the main quantum numbers assigned to electrons are the principal quantum number (n), angular momentum. the quantum numbers are parameters that describe the distribution of electrons in the atom, and therefore its fundamental.. What Are The 4 Types Of Quantum Numbers.
From daphne-has-burnett.blogspot.com
What Are the 4 Quantum Numbers Describe Each What Are The 4 Types Of Quantum Numbers in atoms, there are a total of four quantum numbers: there are four quantum numbers that we are going to discuss: The principal quantum number (n), the orbital angular momentum quantum number (l), the magnetic. the four quantum numbers. there are four quantum numbers, namely, principal, azimuthal, magnetic and spin quantum numbers. This is the same. What Are The 4 Types Of Quantum Numbers.